Quick answer: The famous "26% increase in breast cancer risk" from the 2002 WHI press release was a relative risk; the absolute risk was about 8 extra cases per 10,000 women per year on combined HRT after 5+ years of use. Estrogen-only HRT (for women who had a hysterectomy) actually showed a decrease in breast cancer in the same trial — about 7 fewer cases per 10,000 per year — a result rarely mentioned. The progestin matters more than the estrogen: bioidentical micronized progesterone shows lower risk in observational data than the synthetic medroxyprogesterone used in WHI. Short-course HRT (1-3 years) for vasomotor symptoms shows minimal signal. The HRT and breast cancer risk conversation is much more nuanced than the headline.
The 60-second version
If "HRT causes breast cancer" is what stopped you, read this
You searched HRT breast cancer risk at midnight because you have hot flashes that wreck your sleep, brain fog that's affecting your work, vaginal dryness that's affecting your marriage, and joint pain that woke you up again — and someone, somewhere, told you HRT was off the table because of breast cancer. Maybe your mother died of breast cancer in the 90s and your aunt lectured you. Maybe your primary-care doctor mentioned it in passing as if it were obvious. Maybe you read the 2002 JAMA headline yourself: "Combined HRT increases breast cancer risk by 26 percent." That number became cultural canon. It almost single-handedly drove HRT prescription rates down 80% over the decade that followed. It also was, in absolute terms, much smaller than the headline made it sound.
This article is the part most clinicians don't have time to walk through in a 12-minute appointment. It is the honest absolute-risk math (the actual case numbers, not the relative percentages), the WHI estrogen-only arm result that almost no public conversation includes, the bioidentical progesterone distinction that the 2002 trial couldn't even test (because the trial only used medroxyprogesterone acetate), and a side-by-side comparison of how HRT and breast cancer risk stacks up against other modifiable risk factors most women already accept without a second thought. None of this is HRT cheerleading. None of it is "WHI was wrong." It is the precision the conversation has been missing.
The HRT and breast cancer risk conversation has three layers, and the public discussion mostly skipped two of them.
Layer 1: Absolute risk vs relative risk. WHI's combined-HRT arm produced a 26% relative increase, which sounds large. In absolute terms it was approximately 8 extra cases of invasive breast cancer per 10,000 women per year — roughly 1 extra case per 1,250 women treated annually. The same magnitude as drinking two alcoholic drinks per day or carrying a postmenopausal BMI of 30+.
Layer 2: The estrogen-only arm reduced breast cancer. WHI's estrogen-only arm (for women with hysterectomy) showed a decrease in breast cancer of about 7 fewer cases per 10,000 women per year (Anderson 2012, Lancet Oncol; Manson 2017, JAMA). This rarely makes the news cycle.
Layer 3: The progestin matters more than the estrogen. WHI used medroxyprogesterone acetate (MPA), a synthetic progestin. Fournier et al.'s 2008 French E3N cohort (~80,000 women) found combined therapy with bioidentical micronized progesterone was not associated with a statistically significant breast cancer increase over the first five years, while MPA-containing combinations were.
Citations: Chlebowski RT, et al. Estrogen plus progestin and breast cancer incidence and mortality in postmenopausal women. JAMA. 2010;304(15):1684-1692. PMID: 20460630 · Anderson GL, et al. Conjugated equine oestrogen and breast cancer incidence and mortality in postmenopausal women with hysterectomy: extended follow-up of the WHI randomised placebo-controlled trial. Lancet Oncol. 2012;13(5):476-486. PMID: 22356633 · Manson JE, et al. Menopausal hormone therapy and long-term all-cause and cause-specific mortality: WHI Randomized Trials. JAMA. 2017;318(10):927-938. PMID: 28898378 · Fournier A, et al. Unequal risks for breast cancer associated with different hormone replacement therapies: results from the E3N cohort study. Breast Cancer Res Treat. 2008;107(1):103-111. PMID: 17924148.
What the WHI study actually found
The Women's Health Initiative was the largest randomized trial of hormone therapy ever conducted in postmenopausal women. It enrolled approximately 27,000 women in two parallel arms — one comparing combined estrogen-plus-progestin (conjugated equine estrogens 0.625 mg + medroxyprogesterone acetate 2.5 mg, marketed as Prempro) against placebo in women with an intact uterus, and a second comparing estrogen-alone (conjugated equine estrogens 0.625 mg, marketed as Premarin) against placebo in women who had previously had a hysterectomy. The combined arm was stopped early in July 2002 after the data safety monitoring board flagged a breast cancer signal. The press release that followed reported the now-famous 26% relative increase in invasive breast cancer in the combined-HRT arm.
Here is what the press release didn't lead with. The combined-HRT signal was small in absolute terms. The placebo arm produced approximately 30 cases of invasive breast cancer per 10,000 women per year. The combined-HRT arm produced approximately 38 cases — about 8 extra cases per 10,000 women per year, or roughly 1 extra case per 1,250 women treated for a year. Chlebowski et al.'s 2010 follow-up paper in JAMA, looking at the full WHI cohort with extended follow-up, refined this number and confirmed it accumulated with longer duration of use, with the signal becoming more visible after about 5 years of combined therapy.
Now here is the part the public conversation collapsed. The estrogen-only arm — same trial, same researchers, same statistical methods — produced a different result. Anderson et al.'s 2012 paper in Lancet Oncology, the long-term follow-up of the estrogen-only arm, reported that women on estrogen-only therapy had a lower rate of invasive breast cancer than women on placebo: an absolute reduction of approximately 7 fewer cases per 10,000 women per year, statistically significant. Manson et al.'s 2017 long-term mortality follow-up in JAMA confirmed the breast cancer mortality benefit in the estrogen-only arm was sustained over an 18-year follow-up window. The estrogen-only result was reported. It just never went viral the way the combined-arm result did.
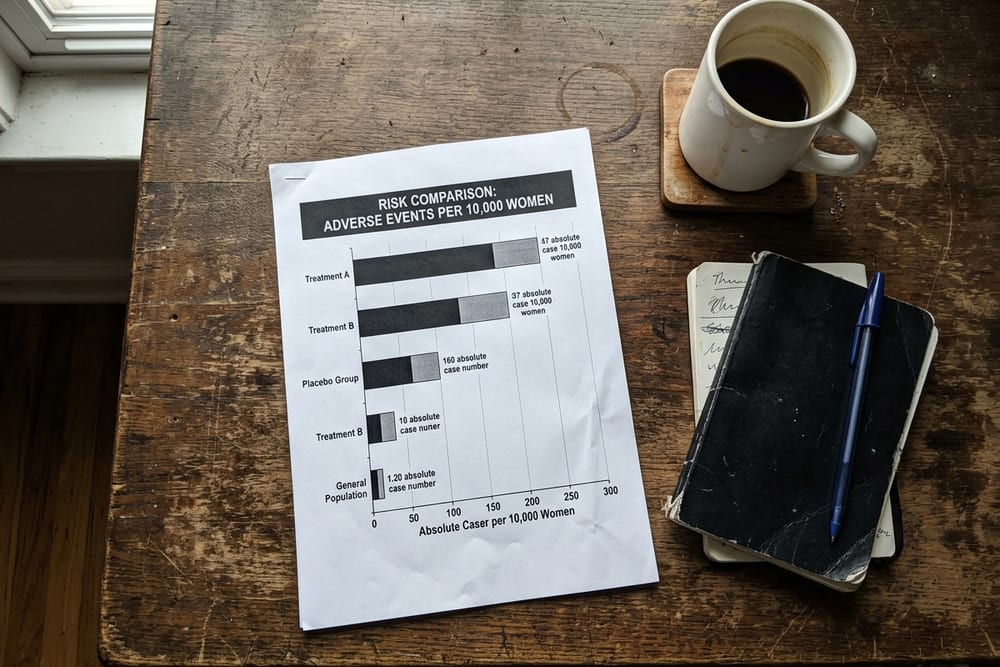
The clinical takeaway from WHI is not that HRT causes breast cancer. The takeaway is more specific: combined estrogen-plus-medroxyprogesterone for 5+ years is associated with about 8 extra cases of invasive breast cancer per 10,000 women per year. Estrogen alone, in the WHI population, was not — it was associated with a small decrease. The conflation of "HRT" into one undifferentiated category, and the foregrounding of the relative risk over the absolute risk, are both reasons the public conversation about HRT and breast cancer risk has been distorted for two decades. The numbers are knowable. They have always been knowable. They simply haven't been the part anyone repeated.
It is also worth noting that the average WHI participant was 63 years old at enrollment — well past peak menopause and not the typical population a clinician today would start on HRT. Subsequent re-analyses (Manson et al. 2013, 2017) developed what is now called the "timing hypothesis": breast cancer signals, cardiovascular signals, and overall mortality signals are different in women who start HRT closer to menopause onset (roughly under age 60, within 10 years of menopause) versus women who start later. The current NAMS 2022 Position Statement and the 2024 update of the Endocrine Society's Menopause and Hormone Therapy Clinical Practice Guideline both incorporate the timing hypothesis explicitly, and both are more permissive about HRT initiation in symptomatic women under 60 than the post-2002 prescriber's reflex would suggest.
The progestin distinction (this is the part the trial couldn't test)
Here is the molecular detail that didn't make it into the headlines. The combined-HRT arm of WHI used a single progestin: medroxyprogesterone acetate, abbreviated MPA. MPA is a synthetic progestin, structurally derived from progesterone but modified to give it oral activity and a longer half-life. The modification also changes its receptor-binding profile — MPA has weak androgenic activity, distinct effects on the progesterone receptor, and (relevant to this conversation) a different effect on breast tissue than bioidentical progesterone. WHI did not test bioidentical progesterone. It tested MPA. The 26% relative-risk headline applied specifically to estrogen-plus-MPA — but in the years that followed, the public conversation generalized it to all progestins, including bioidentical micronized progesterone, which has a fundamentally different molecular structure.
Fournier et al.'s 2008 paper in Breast Cancer Research and Treatment is the largest dataset addressing this distinction. The French E3N cohort followed approximately 80,000 women over a median of 8 years. Combined therapy with synthetic progestins (including MPA) showed a statistically significant increase in breast cancer risk — consistent with WHI. Combined therapy with bioidentical micronized progesterone (the formulation marketed as Prometrium in the US, Utrogestan in Europe) showed no statistically significant increase over the first 5 years of use, with a smaller and not-quite-statistically-significant signal at longer durations. The effect-size difference was clinically meaningful: synthetic progestins roughly doubled the magnitude of the breast cancer signal compared with micronized progesterone.
The mechanism is not yet fully resolved, but the leading explanations are: (1) MPA's androgenic activity and unique progesterone-receptor binding may stimulate breast tissue differently than the bioidentical molecule does; (2) different progestins produce different downstream effects on breast cell proliferation and apoptosis; and (3) different molecules clear the body at different rates and have different tissue accumulation profiles. None of these is settled science at the molecular level, but the epidemiologic distinction in Fournier 2008 has been replicated in subsequent observational work, and both the NAMS 2022 Position Statement and the 2024 Endocrine Society guideline now reference it as a clinically relevant consideration when choosing the progestin component of combined HRT.
What this means for HRT and breast cancer risk: if you have a uterus and need progestin to protect the endometrium, the type of progestin you choose has implications. Bioidentical micronized progesterone (taken at bedtime, typically 100-200 mg) is not categorically risk-free, but the observational evidence suggests its breast cancer signal is smaller than the synthetic-progestin signal that drove the WHI headline. RCT-level evidence comparing bioidentical progesterone head-to-head with MPA on breast cancer endpoints has not been published — that trial would take 10+ years and tens of millions of dollars and is unlikely to happen — so the recommendation rests on Fournier 2008 and confirmatory observational studies. Our bioidentical vs synthetic HRT explainer walks through the rest of the molecule-by-molecule comparison.

The duration math: short-course vs long-term
The HRT and breast cancer risk conversation also gets foreshortened in another important way: it almost never separates short-course therapy (1-3 years for vasomotor symptoms) from long-term therapy (5+ years). The WHI signal emerged at roughly 5 years of combined HRT use; the absolute risk increase was estimated at approximately 8 extra cases per 10,000 women per year, but that average distribution was not uniform across years 1 through 7. Years 1-2 of combined HRT in WHI showed minimal additional breast cancer signal. Years 3-4 began to show a small accumulating signal. Years 5+ produced the bulk of the headline number.
For a woman starting HRT specifically for severe vasomotor symptoms (hot flashes, night sweats), with the plan to taper after 1-3 years, the breast cancer math is meaningfully smaller than the WHI headline implied. NAMS 2022 explicitly notes this distinction, and recommends that the duration of HRT be matched to the duration of need rather than a fixed maximum. The Endocrine Society 2024 guideline update echoes the same principle. The "shortest duration possible" framing of the early 2000s — which often translated into 6-12 month maximums — has been replaced with individualized duration based on symptom severity, ongoing benefit, and personal risk profile.
This matters clinically because women considering HRT for severe symptoms are often making the decision in their late 40s or early 50s. The mortality benefit of treating moderate-to-severe vasomotor symptoms — improved sleep, lower fall risk, better bone density, reduced osteoporotic fracture incidence — accumulates immediately. The breast cancer signal accumulates over years, and is small in absolute terms even at the long-term tail. Our companion piece on HRT side effects covers the broader trade-off picture, including the ones that show up in the first weeks (and the ones that don't show up at all in most women).
The other-risk-factors comparison (this is the lens most articles miss)
Here is the framing question that almost no breast-cancer-and-HRT article asks: compared to what? A woman weighing HRT and breast cancer risk is not making a decision in a vacuum. She is making it against a baseline that already includes other modifiable risk factors — alcohol, weight, exercise — that operate on the same breast cancer biology, often at similar magnitudes, and that almost no one talks about with the same urgency as HRT.
Below are the absolute risk magnitudes for several common breast cancer risk factors, expressed in the same currency as the HRT signal: extra cases per 10,000 women per year. The numbers are approximate and derived from major epidemiologic reviews; the point is the relative scale, not the third decimal point.
Look at that scale. The BRCA1/2 magnitude is orders of magnitude larger than every other factor — that is its own clinical category, and women who carry those mutations are managed by specialty oncology, not primary care. Below that, the magnitudes cluster: family history adds the most among non-genetic factors; combined HRT, daily alcohol, and obesity all add roughly similar magnitudes (5-10 extra per 10,000 per year); sedentary lifestyle is slightly less; estrogen-only HRT in the hysterectomy population goes the other direction.
The lens this gives you: combined HRT for 5+ years adds approximately the same absolute breast cancer risk as drinking two glasses of wine every night does. We do not tell women not to drink wine because of breast cancer risk. We tell them about it, we let them make the call, and we do not pretend the decision is morally weighted differently than it really is. HRT has been held to a different standard, and the absolute-risk math is one of the reasons that standard is starting to shift.
None of this is to say breast cancer risk is irrelevant. It is to say breast cancer risk is one input into a larger personal calculation that also includes severity of menopausal symptoms, fracture risk, cardiovascular risk, quality of life, and personal risk factors like family history. The math has to add up to a defensible decision for you, in your specific picture — not as a yes/no answer to a single question.
What this means for your decision
Translating absolute-risk math into your own decision goes one of three ways, depending on your situation. None of these is a substitute for an actual clinical conversation with someone who knows your full history, but the framework is useful.
If you've had a hysterectomy
You don't need progestin (because progestin's job is to protect the endometrium, and you no longer have one). Your HRT option is estrogen-only, and the WHI estrogen-only arm result is the directly relevant data point: net decrease of about 7 fewer cases of invasive breast cancer per 10,000 women per year. The breast cancer math, in your case, is favorable for HRT, not unfavorable. The clinical conversation focuses on cardiovascular risk, stroke risk, and symptom severity — not breast cancer. This is the population for whom the most common HRT objection ("but breast cancer") doesn't actually apply on the data.
If you have an intact uterus
You need progestin alongside estrogen to protect the endometrium, and the choice of progestin is where the conversation lives. Combined HRT with synthetic progestin (medroxyprogesterone) for 5+ years carries the WHI signal: about 8 extra cases of breast cancer per 10,000 women per year. Combined HRT with bioidentical micronized progesterone shows a smaller signal in observational data (Fournier 2008), and most current prescribers default to bioidentical progesterone unless there is a specific reason to use a synthetic progestin. Short-course HRT (1-3 years for severe vasomotor symptoms) shows minimal additional breast cancer signal regardless of which progestin is used. The breast cancer math, in your case, is small and modifiable by progestin choice and duration.
If you have a personal history of breast cancer
Systemic HRT is generally not recommended for women with a personal history of breast cancer. The HABITS trial (Holmberg 2008) was stopped early after showing increased recurrence risk in breast cancer survivors who started HRT, and major guidelines (NAMS, ASCO, ACOG) treat prior breast cancer as a relative-to-absolute contraindication for systemic therapy. For severe vasomotor symptoms in this population, non-hormonal options are first-line: SSRIs (paroxetine), SNRIs (venlafaxine), gabapentin, clonidine, and the newer neurokinin-3-receptor antagonist fezolinetant. Vaginal estrogen for genitourinary symptoms is treated as a separate clinical decision and is often considered acceptable after specialty consultation — the systemic absorption from low-dose vaginal preparations is minimal, and major bodies (NAMS, ACOG, ASCO) permit it in survivors when non-hormonal options have failed. This decision is patient-by-patient, oncology-coordinated, and carefully documented; it is not a self-service judgment call.
If you have a strong family history but no personal cancer history
This is the most common nuanced case. A first-degree relative with breast cancer (mother, sister, daughter) raises baseline risk by roughly 1.5-2×, which means the same 8-per-10,000 absolute increase from combined HRT becomes a slightly larger absolute number against an already-higher baseline. Known BRCA1/BRCA2 mutations are a much stronger contraindication and warrant specialty consultation before any HRT discussion. For women with a moderate family history but no known mutation, many clinicians still prescribe HRT — typically estrogen-only if hysterectomy, or estrogen plus bioidentical micronized progesterone if uterus — when symptoms are severe, but the conversation is longer and includes baseline mammography, careful annual review, and informed-consent documentation. Our explainer on when to start HRT walks through the timing-hypothesis logic that interacts with this decision.

How ClearedRx handles HRT and breast cancer risk in practice
ClearedRx is a doctor-supervised HRT service for women, online. You take a one-minute quiz. A licensed physician in our network reviews your symptoms and history within 24 hours. If you are a fit, they prescribe — and your treatment ships discreetly to your door, the same week. We prescribe both compounded and FDA-approved HRT preparations; the patient picks based on cost, format preference, and clinical fit. Our flagship preparation, the compounded estrogen + progesterone vaginal cream, uses bioidentical micronized progesterone — not medroxyprogesterone — which means the progestin choice is already aligned with the lower-signal Fournier 2008 evidence base.
The intake covers personal and family breast cancer history, prior screening mammography, and personal risk factors (family history, BRCA status if known, prior atypical biopsies, etc.). Women with a personal history of breast cancer, known BRCA1/BRCA2 mutations, or recent (within 12 months) abnormal mammograms are routed to specialist consultation rather than direct HRT initiation; this is medical caution rather than a sales pitch. Women with moderate family history and severe symptoms are typically prescribed a regimen that uses estrogen-only (if hysterectomy) or estrogen plus bioidentical progesterone (if uterus), with annual mammography baked into the treatment plan and clear informed-consent documentation.
Cost framing: ClearedRx HRT starts at $49 per month for compounded preparations and $89 per month for FDA-approved generics, all-in (medication, doctor reviews, free shipping in all 50 states). New patients receive 50% off their first month. There are no surprise fees and no insurance paperwork. For broader cost context across formulations, our HRT cost comparison walks through every preparation across every channel.

"The Women's Health Initiative findings on hormone therapy and breast cancer risk are most appropriately interpreted in absolute terms. The combined-therapy arm produced approximately 8 extra cases of invasive breast cancer per 10,000 women per year, while the estrogen-alone arm in women with prior hysterectomy showed a reduction in breast cancer incidence. Type of progestin, duration of therapy, and timing of initiation all modify these estimates." — The North American Menopause Society. The 2022 Hormone Therapy Position Statement. Menopause. 2022;29(7):767-794.
If you also want to map the rest of the picture
The HRT and breast cancer risk conversation is one input into a larger menopause-management picture. Our free Menopause Symptom Score is a 60-second self-check that scores your symptom cluster as a single hormonal-fingerprint number, which is often the most useful starting point before the breast-cancer-risk conversation. For the full symptom catalogue, see our 34 symptoms of perimenopause piece. For the broader risk-benefit framing, our signs you need HRT piece walks through when the conversation is worth having, and our when to start HRT piece covers the timing-hypothesis logic that interacts with breast cancer risk. Our bioidentical vs synthetic HRT explainer covers the progestin distinction in detail. Our HRT side effects piece covers the broader side-effect profile (the ones that show up in the first weeks, and the ones that don't), and our HRT cost comparison walks through pricing across every formulation. For prevalence numbers across the menopause symptom landscape, the menopause statistics 2026 page has the data.
Frequently asked questions
Does HRT cause breast cancer?
Combined HRT (estrogen plus a synthetic progestin) was associated with about 8 extra cases of breast cancer per 10,000 women per year in the WHI trial after 5+ years of use — a small absolute increase that produced the famous "26% relative risk" headline. Estrogen-only HRT in women with hysterectomy actually showed a decrease in breast cancer risk in the same trial — about 7 fewer cases per 10,000 women per year. The progestin used in WHI (medroxyprogesterone) drove most of the combined-arm signal; bioidentical micronized progesterone shows lower risk in observational data (Fournier 2008). Short-course HRT (1-3 years) shows minimal signal.
What is the absolute risk of breast cancer on HRT?
On combined HRT, baseline breast cancer incidence in postmenopausal women is roughly 33 cases per 10,000 women per year. Combined HRT for 5+ years raises that to about 41 per 10,000 — an additional 8 cases per 10,000, or roughly 1 extra case per 1,250 women per year. For estrogen-only HRT in women with hysterectomy, WHI found about 7 fewer cases per 10,000 per year over a median 7+ years of follow-up. These absolute numbers are how clinicians actually weigh the risk-benefit tradeoff against menopausal symptom severity, fracture-prevention benefit, and quality-of-life impact.
Is bioidentical progesterone safer than synthetic progestins for breast cancer risk?
Observational evidence suggests yes, though randomized-trial-level evidence is still limited. Fournier et al.'s 2008 French E3N cohort study followed roughly 80,000 women and found combined therapy with bioidentical micronized progesterone was associated with no statistically significant increase in breast cancer risk over the first five years, while synthetic progestins (including medroxyprogesterone) showed a statistically significant increase. The proposed mechanism: micronized progesterone has a different molecular structure and binding profile to progesterone receptors and avoids the androgenic activity of medroxyprogesterone. NAMS and the Endocrine Society now both note this distinction in guidance documents, though they stop short of declaring micronized progesterone definitively safer until RCT-level data is available. See our bioidentical vs synthetic HRT explainer for the full breakdown.
Does estrogen-only HRT increase breast cancer?
No — and this is the result almost no one talks about. The WHI estrogen-only arm (for women who had a hysterectomy) showed a decrease in breast cancer incidence, not an increase. Anderson et al.'s 2012 Lancet Oncology paper reported an absolute reduction of approximately 7 fewer cases per 10,000 women per year, and the long-term follow-up by Manson et al. (2017, JAMA) confirmed the breast cancer mortality benefit was sustained. The headline "26% increase" applied only to the combined estrogen-plus-progestin arm. The estrogen-only arm result was never given proportional press attention, which is one reason public perception of HRT and breast cancer risk is so distorted.
How long is HRT safe to take?
The breast cancer signal in WHI emerged after about 5 years of combined HRT use, was small in absolute terms (about 8 extra cases per 10,000 women per year), and accumulated with longer duration. Short-course HRT (1-3 years) targeted at vasomotor symptoms shows minimal additional breast cancer risk. Most current guidelines (NAMS 2022, Endocrine Society 2024 update) recommend reassessing the risk-benefit conversation annually but do not impose a hard duration cap. The "shortest duration possible" framing of the early 2000s has been replaced with "individualized duration based on symptom severity, ongoing benefit, and personal risk profile."
Can I take HRT if I have a family history of breast cancer?
Family history alone does not automatically rule out HRT, but it shifts the risk-benefit conversation. A first-degree relative with breast cancer (mother, sister, daughter) raises baseline risk by roughly 1.5-2×, which means the same 8-per-10,000 absolute increase from combined HRT becomes a slightly larger absolute number against an already-higher baseline. Known BRCA1/BRCA2 mutations are a much stronger contraindication and warrant specialty consultation. For women with a moderate family history but no known mutation, many clinicians will still prescribe HRT (especially estrogen-only or estrogen plus bioidentical progesterone) when symptoms are severe — but the conversation gets longer and includes baseline mammography, careful annual review, and informed-consent documentation.
Can I take HRT after breast cancer?
Generally no for systemic HRT — a personal history of breast cancer is widely treated as a relative or absolute contraindication to systemic estrogen and progestin therapy. The HABITS trial (Holmberg 2008) showed an increased recurrence risk in breast cancer survivors who started HRT, and the trial was stopped early. For severe vasomotor symptoms after breast cancer, non-hormonal options (SSRIs, SNRIs, gabapentin, clonidine, fezolinetant) are first-line. Vaginal estrogen for genitourinary symptoms is treated separately and is often considered acceptable in many breast cancer survivors after specialty consultation, because systemic absorption from low-dose vaginal preparations is minimal — but this decision is patient-by-patient and oncology-coordinated. Do not start systemic HRT after breast cancer without an oncology-coordinated conversation.
Is vaginal estrogen safe after breast cancer?
Low-dose vaginal estrogen is treated as a separate clinical decision from systemic HRT and is often considered acceptable in breast cancer survivors with severe genitourinary symptoms (vaginal dryness, dyspareunia, recurrent UTIs) after coordination with their oncology team. NAMS, ACOG, and the American Society of Clinical Oncology all permit low-dose vaginal estrogen in breast cancer survivors when non-hormonal options have failed, citing minimal systemic absorption. The decision is individualized — particularly for women on aromatase inhibitors — and requires shared decision-making between the patient, the oncologist, and the prescribing clinician.
Why did WHI cause so much HRT confusion?
Three reasons. First, the press release led with the relative risk ("26% increase") rather than the absolute risk (about 8 extra cases per 10,000 women per year), which made the signal sound much larger to lay readers than the underlying math justified. Second, the public conversation collapsed both arms of WHI ("combined" and "estrogen-only") into one undifferentiated "HRT causes breast cancer" message, even though the estrogen-only arm actually showed a decrease. Third, the average WHI participant was 63 years old at enrollment — well past peak menopause and not the typical population a clinician would start on HRT today. Subsequent re-analyses (Manson 2013, 2017) showed the cardiovascular and breast cancer signals are different in women who start HRT closer to menopause onset (the "timing hypothesis").
Should I get a baseline mammogram before starting HRT?
Yes — a baseline mammogram before initiating combined HRT (and annual mammograms while on therapy) is standard practice and is recommended in NAMS, ACOG, and Endocrine Society guidance. The point is not that HRT will cause a new cancer in the first year (it will not), but to make sure no existing breast cancer is missed before adding a hormonal therapy that can stimulate hormone-receptor-positive tumors. If a baseline screening mammogram is overdue, most prescribing clinicians will require it before issuing the prescription — which is good medicine, not gatekeeping.
Sources & references
- Chlebowski RT, Anderson GL, Gass M, et al. Estrogen plus progestin and breast cancer incidence and mortality in postmenopausal women. JAMA. 2010;304(15):1684-1692. PMID: 20460630
- Anderson GL, Chlebowski RT, Aragaki AK, et al. Conjugated equine oestrogen and breast cancer incidence and mortality in postmenopausal women with hysterectomy: extended follow-up of the WHI randomised placebo-controlled trial. Lancet Oncol. 2012;13(5):476-486. PMID: 22356633
- Manson JE, Aragaki AK, Rossouw JE, et al. Menopausal Hormone Therapy and Long-term All-Cause and Cause-Specific Mortality: The Women's Health Initiative Randomized Trials. JAMA. 2017;318(10):927-938. PMID: 28898378
- Fournier A, Berrino F, Clavel-Chapelon F. Unequal risks for breast cancer associated with different hormone replacement therapies: results from the E3N cohort study. Breast Cancer Res Treat. 2008;107(1):103-111. PMID: 17924148
- The North American Menopause Society. The 2022 Hormone Therapy Position Statement of The North American Menopause Society. Menopause. 2022;29(7):767-794. PMID: 35797481
- Stuenkel CA, Davis SR, Gompel A, et al. Treatment of Symptoms of the Menopause: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2015;100(11):3975-4011. PMID: 26444994
- Holmberg L, Iversen OE, Rudenstam CM, et al. Increased risk of recurrence after hormone replacement therapy in breast cancer survivors. J Natl Cancer Inst. 2008;100(7):475-482. PMID: 18364505
- Manson JE, Chlebowski RT, Stefanick ML, et al. Menopausal hormone therapy and health outcomes during the intervention and extended poststopping phases of the Women's Health Initiative randomized trials. JAMA. 2013;310(13):1353-1368. PMID: 24084921
- Internal: HRT side effects · bioidentical vs synthetic HRT · HRT cost comparison · when to start HRT · signs you need HRT · 34 symptoms of perimenopause · menopause statistics 2026 · menopause symptom score tool
Talk to a clinician about HRT and your specific breast cancer risk profile
ClearedRx prescribes evidence-based HRT — bioidentical estradiol, micronized progesterone, or a combined cream — based on your full picture, with 24-hour MD review and free shipping in all 50 states. Compounded preparations from $49/month, FDA-approved generics from $89/month. No insurance hassle. New patients save 50% on the first month.
Find out if HRT is right for me



